 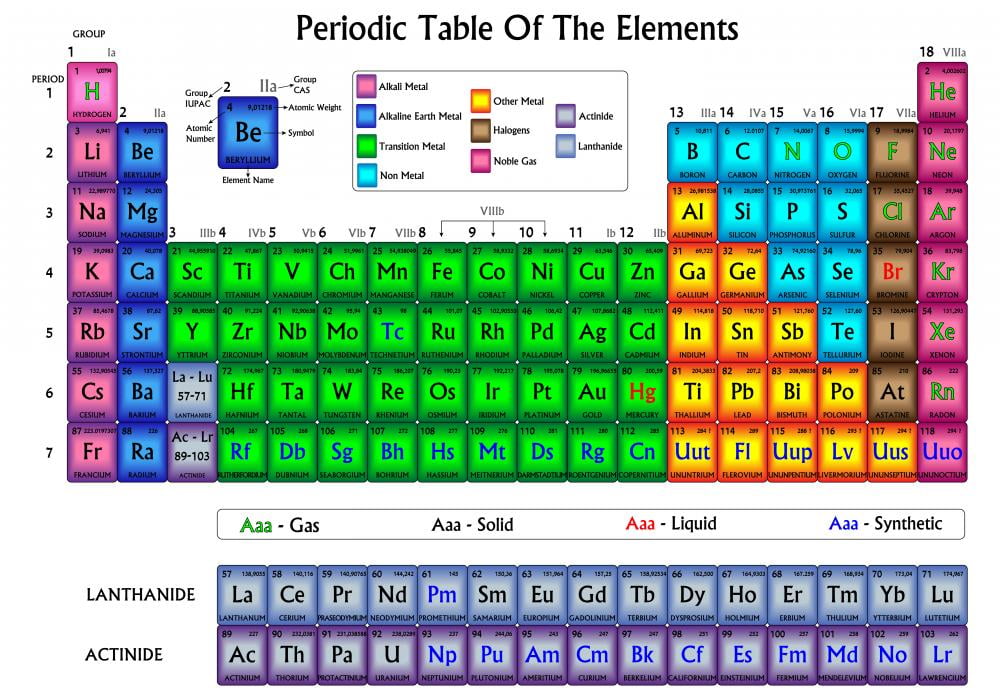
The rare earths are silver, silvery-white, or gray metals.Helium (He) has only 2 electrons in the outer shell = Full.Very reactive are often bonded with elements from Group 1.Contains metals, metalloids, and non-metals.Contains metals, metalloids, and a non-metal Carbon (C).Can bond with many elements in a variety of shapes.The transition metals are able to put up to 32 electrons in their second to last shell.Most periodic tables are color-coded to provide extra information about parts and element communities. Good conductors of heat and electricity. Highest periodic desks belong color-coded to offering added related about elements and factor groups.Hydrogen is not a member, it is a non-metal.Elements in each family react differently with other elements.Group numbers above each column (1 18) To print your own, here is a zip file of jpeg photos that you can upload or bring to your photo printer: Periodic Table Images ZIP file. Each family has a specific name to differentiate it from the other families in the periodic table. Color-coded background for element families/groups Symbols in black (solid), blue (liquid), or red (gas) elements (at room temp.) Synthetic elements are in gray.The horizontal rows on the periodic table are called periods. All the members of a family of elements have the same number of valence electrons and similar chemical properties. Elements on the periodic table can be grouped into families bases on their chemical properties. The vertical columns on the periodic table are called groups or families because of their similar chemical behavior.Reactivity Alkali Metals and Halogens are very reactive because they have either 1 or 7 valence electrons Boron Group or Earth Metals: Group 13 (IIIA) 3 valence electrons. Transition Metals: Groups 3-12 d and f block metals have 2 valence electrons. When you look at a periodic table, each of the rows is considered to be a different period Under this classification system, element families correspond to their periodic table column, which in turn reflects their typical number of valence electrons. The elements in a group have the same number of electrons in their outer orbital (Valence). Period and Groups When a column goes from top to bottom, it's called a group. Bean, Discovery Scienceĭirections Obtain a periodic table or download it Read through the PowerPoint and color as indicated Answer post-project questions sheet. Coloring Part 1 Coloring the Periodic Table Families Mr. Vocabulary and Questions about the periodic table including groups, periods, families and basic trends.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
